Douglas Melton’s laboratory at the Harvard University Stem Cell Institute in Cambridge, Massachusetts has discovered a liver hormone that stimulates the growth of insulin-secreting beta cells in the pancreas. This discovery could very well lead to new treatments for diabetes.
This hormone, betatrophin, was induced in mice by treating them with a peptide that binds to insulin receptors. The insulin-occupied insulin receptors were unable to bind insulin, and that caused the animals to be resistant to insulin. Under these conditions, the livers of these mice produced betatrophin, which caused the animals’ insulin-secreting pancreatic β cells to proliferate. Melton and others searched for genes that showed increased activity under these insulin-resistant conditions, which allowed Melton and colleagues to isolate and identify betatrophin.
According to Melton and his co-workers, “Transient expression of betatrophin in mouse liver significantly and specifically promotes pancreatic β cell proliferation, expands β cell mass, and improves glucose tolerance” (from the abstract of the paper).
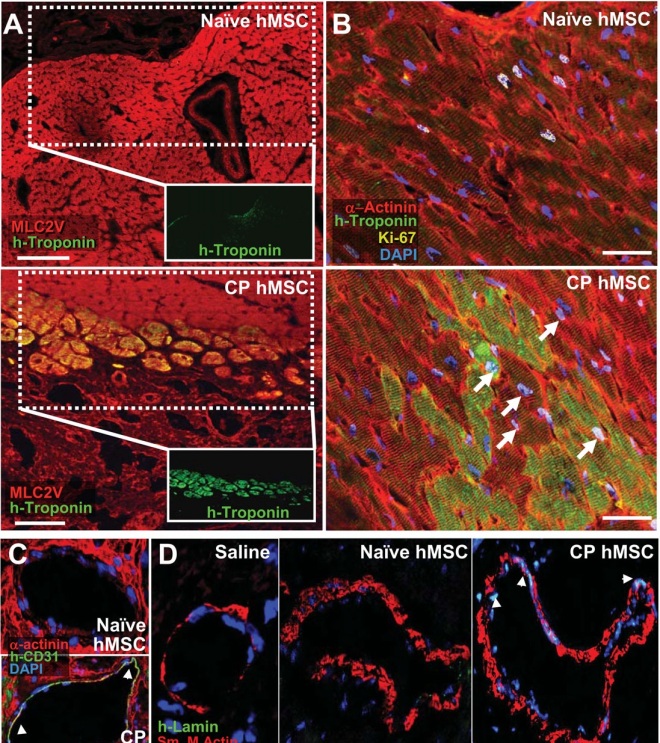
Further experiments showed that when eight-week-old mice injected with betatrophin there was an average 17-fold rise in the proliferation of their insulin-secreting pancreatic β cells. Melton and others published these results in the journal Cell. Fortunately, betatrophin is also found in the human liver, according to Melton and others.
“It’s rare that one discovers a new hormone, and this one is interesting because it’s so specific,” says Melton. “It works only on β cells and it’s so robust and so potent.”
Pancreatic β cells replicate rapidly during embryonic and neonatal stages in both mice and humans, but beta cell growth decreases dramatically in adults. A decrease in the function of beta cells late in life is the main cause of type 2 diabetes. Type 2 diabetes is a metabolic disorder that affects more than 300 million people worldwide. In the United States alone, the two forms of diabetes — type 2 and type 1— account for US$176 billion in direct medical costs each year.
Melton hypothesized that injections of betatrophin once a month, or even once a year, could potentially induce enough activity in pancreatic β cells to provide the same level of blood-sugar control for people with type 2 diabetes as do daily insulin injections. According to Melton, betatrophin would cause fewer complications, since the body would make its own insulin. He also hopes that betatrophin will be able to help people with type 1 diabetes.
Matthias Hebrok, director of the University of California, San Francisco, Diabetes Center, says that the work “is a great advance”. “The findings are very interesting,” he says, although he would like to see the experiments repeated in older mice. “Do mice that are on their way to becoming diabetic at an advanced age truly have an increase in proliferative capacity upon treatment with betatrophin?” he asks. This is a fair question.
Henrik Semb, managing director of the Danish Stem Cell Center in Copenhagen, says that “the identification of a factor, betatrophin, that stimulates mouse β-cell replication with remarkable efficiency is a very important discovery, because it provides the starting point for further studies to elucidate the underlying mechanism of β-cell replication.”
β-cell replication has proved difficult to control in humans, but producing enough betatrophin for testing in human clinical trials will take about two years, according to Melton, who is also working to identify the hormone’s receptor and its mechanism of action.
References: Yi, P., Park, J.-S. & Melton, D. A. Cell http://dx.doi.org/10.1016/j.cell.2013.04.008 (2013).